The Most Popular Contraceptive Drugs
The Most Popular Contraceptive Drugs

The Most Famous Drugs Most Commonly used as Contraceptives are:

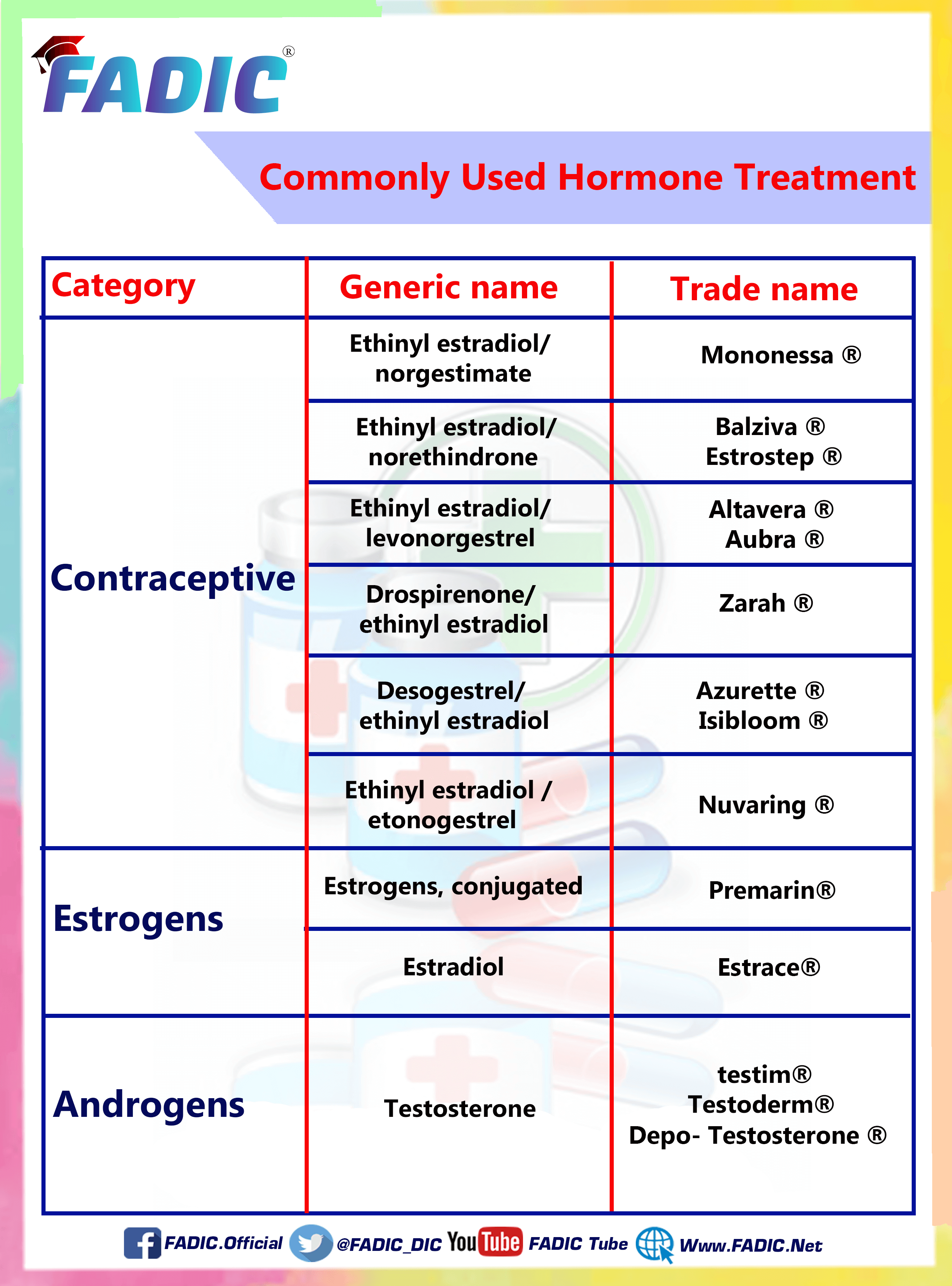
Ethinyl Estradiol and Desogestrel

- Brand Names: Ortho-Cept
- Therapeutic Category: Contraceptive, Estrogen and Progestin Combination
- Dosage Forms: Tablet, Oral
- Use: Labeled Indications:
- Contraceptive
- Off Label: Polycystic ovary syndrome, Menstrual bleeding, Hirsutism, Dysmenorrhea, Pain associated with endometriosis, Abnormal uterine bleeding
- Dosing : Adult: Importantly, Contraceptive Drugs: Oral: 1 tablet once daily
- Dose Adjustments
- Renal Impairment: no dosage adjustments
- Hepatic Impairment: contraindicated in patients with hepatic dysfunction.
- Adverse Drug Interaction:
Depression, headache, migraine, mood changes, fluid retention, weight gain, breast tenderness, vaginal discharge - Pharmacodynamics/Kinetics:
- First, Absorption: Desogestrel and Ethinyl estradiol: Rapid and complete
- Secondly, Metabolism: Hepatic
- Thirdly, Bioavailability: Monophasic preparations: Etonogestrel: 84%; Ethinyl estradiol: 83%
- Then, Half-life elimination: Etonogestrel: 38 ± 20 hours; Ethinyl estradiol: 26 ± 6.8 hours
- Time to peak: Etonogestrel: 1.4 ± 0.8 hours; Ethinyl estradiol: 1.5 ± 0.8 hours
- Important Notes:
- Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptives use. In addition to, this risk increases with age, particularly in women over 35 years of age.
- Furthermore, the number of cigarettes smoked.
- Moreover, for this reason, combination oral contraceptives should not be used by women over 35 who smoke.
- Pregnancy & Lactation: contraindicated in pregnant women/ may be present in breast milk
- Medication Safety issue:
- Firstly, Apri may be confused with Apriso
- Secondly, Ortho-Cept may be confused with Ortho-Cyclen

Ethinyl Estradiol and Drospirenone

- Brand Names: Yasmin
- Therapeutic Category: Contraceptive Drugs, Estrogen and Progestin Combination
- Dosage Forms: Tablet, Oral
- Use: Labeled Indications:
- Contraceptive, Acne vulgaris
- Off Label: Polycystic ovary syndrome, Menstrual bleeding, Hirsutism, Dysmenorrhea, Pain associated with endometriosis, Abnormal uterine bleeding
- Dosing: Adult : Contraception: Oral: 1 tablet once daily
- Dose Adjustments
- Renal Impairment: contraindicated in patients with renal dysfunction.
- Hepatic Impairment: contraindicated in patients with hepatic dysfunction.
- Adverse Drug Interaction:
Arterial thromboembolism, cerebral thrombosis, hypertension, Cerebral haemorrhage. - Moreover, oedema and worsening of varicose veins.
- In addition to exacerbation of systemic lupus erythematosus, Hepatic adenoma, hepatic neoplasm, Abdominal cramps, bloating
- Pharmacodynamics/Kinetics:
- Firstly, Bioavailability: Drospirenone: 76%; Ethinyl estradiol: 40%
- Secondly, Half-life elimination: Terminal: Drospirenone: 30 hours; Ethinyl estradiol: 24 hours
- Thirdly, Time to peak: 1 to 2 hours
- Important Notes:
- Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptives use. In addition to, this risk increases with age, particularly in women over 35 years of age.
- Furthermore, the number of cigarettes smoked.
- Moreover, for this reason, combination oral contraceptives should not be used by women who are over 35 years of age and smoke. over 35 years of age who
- Pregnancy & Lactation: contraindicated in pregnant women/ present in breast milk
- Medication Safety issue:
- Interestingly, Yaz may be confused with Beyaz, Yasmin

Ethinyl Estradiol and Etonogestrel

- Brand Names: NuvaRing
- Therapeutic Category: Contraceptive Drugs, Estrogen and Progestin Combination
- Dosage Forms: Tablet, Oral
- Use: Labeled Indications:
- Contraceptive
- Off Label: Polycystic ovary syndrome, Menstrual bleeding, Dysmenorrhea, Abnormal uterine bleeding
- Dosing: Adult : Vaginal: One ring, inserted vaginally and left in place continuously for three consecutive weeks.
- Moreover, removed for one week. A new ring is inserted seven days after the last was removed (even if bleeding is not complete).
- In addition, it should be inserted at approximately the same time of day the ring was removed the previous week
- Dose Adjustments
- Renal Impairment: no dosage adjustments
- Hepatic Impairment: contraindicated in patients with hepatic dysfunction.
- Adverse Drug Interaction:
Headache, Intermenstrual bleeding, Vaginitis, Mood changes, Acne vulgaris, Weight gain, amenorrhea, Deep vein thrombosis - Pharmacodynamics/Kinetics:
- Duration: Serum levels (contraceptive effectiveness) decrease after three weeks of continuous use
- Absorption: Ethinyl estradiol, in addition to etonogestrel: Rapid
- Metabolism: Hepatic
- Bioavailability: Vaginal: Ethinyl estradiol: 56% Etonogestrel: 100%
- Half-life elimination: Ethinyl estradiol: 45 hours; Etonogestrel: 29 hours
- Time to peak: Vaginal: Ethinyl estradiol: 59 hours; Etonogestrel: 200 hours
- Important Notes:
- Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptives use. In addition to, this risk increases with age, particularly in women over 35 years of age.
- Moreover, the number of cigarettes smoked.
- For this reason, combination oral contraceptives should not be used by women over 35 who smoke.
- Pregnancy & Lactation: contraindicated in pregnant women/ present in breast milk

Ethinyl Estradiol and Levonorgestrel

- Brand Names: Afirmelle
- Therapeutic Category: Contraceptive Drugs, Estrogen and Progestin Combination
- Dosage Forms: Tablet, Oral
- Use: Labeled Indications:
- Contraceptive, Emergency contraception
- Off Label: Polycystic ovary syndrome, Menstrual bleeding, Hirsutism, Dysmenorrhea, Pain associated with endometriosis
- Dosing: Adult : Contraception: Oral: 1 tablet once daily
- Dose Adjustments
- Renal Impairment: no dosage adjustments
- Hepatic Impairment: contraindicated in patients with hepatic impairment.
- Adverse Drug Interaction:
Arterial thromboembolism, cerebral thrombosis, hypertension, Cerebral haemorrhage. In addition to oedema, worsening of varicose veins. - In addition, the exacerbation of systemic lupus erythematosus
- Pharmacodynamics/Kinetics:
- First, Absorption: Rapid
- Secondly, Metabolism: Hepatic
- Thirdly, Bioavailability: Ethinyl estradiol: 38% to 48%; Levonorgestrel: 100%
- Then, Half-life elimination: Ethinyl estradiol: 12-23 hours; Levonorgestrel: 22-49 hours
- Important Notes:
- Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptives use. In addition, this risk increases with age, particularly in women over 35 years of age, and with the number of cigarettes smoked.
- For this reason, combination oral contraceptives should not be used by women over 35 who smoke.
- Pregnancy & Lactation: Pregnancy Risk Factor X / may be present in breast milk
- Medication Safety issue:
- Firstly, Alesse may be confused with Alecensa
- Secondly, Nordette may be confused with Nicorette
- Thirdly, Portia may be confused with Potiga

Ethinyl Estradiol and Norethindrone

- Brand Names: Alyacen
- Therapeutic Category: Contraceptive Drugs, Estrogen and Progestin Combination
- Dosage Forms: Tablet, Oral
- Use: Labeled Indications:
- Contraceptive, Osteoporosis prevention
- Off Label: Polycystic ovary syndrome, Menstrual bleeding, Dysmenorrhea, Abnormal uterine bleeding, Hirsutism
- Dosing : Adult: Abnormal uterine bleeding, acute (off-label use):
- Moreover, Oral: Ethinyl estradiol 0.035 mg/norethindrone 1 mg three times a day for seven day
- Contraception: Females: One tablet once daily
- Dose Adjustments
- Renal Impairment: no dosage adjustments
- Hepatic Impairment: contraindicated in patients with hepatic dysfunction.
- Adverse Drug Interaction:
Headache, Edema, Change in menstrual flow, Mood changes, Acne vulgaris, Hypertension - Pharmacodynamics/Kinetics:
- Absorption: Rapid
- Bioavailability: 43% to 55%
- Metabolism: Hepatic
- Half-life elimination: 19 to 24 hours
- Important Notes:
- Cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptives use. ّIn addition to, this risk increases with age, particularly in women over 35 years of age.
- Moreover, the number of cigarettes smoked.
- For this reason, combination oral contraceptives should not be used by women over 35 who smoke.
- Furthermore, there is an increased risk of endometrial cancer in a woman with a uterus who uses unopposed estrogens
- Pregnancy & Lactation: Pregnancy Risk Factor X /may be present in breast milk
- Medication Safety Issue:
- Firstly, Femhrt may be confused with Femara
- Secondly, Lo Loestrin Fe may be confused with Loestrin Fe

Ethinyl Estradiol and Norgestimate

- Brand Names: Cyclen
- Therapeutic Category: Contraceptive Drugs, Estrogen and Progestin Combination
- Dosage Forms: Tablet and Oral
- Use: Labeled Indications:
- Treatment of moderate acne vulgaris in females at least 15 years of age, Contraceptive
- Off Label: Polycystic ovary syndrome, Menstrual bleeding, Hirsutism, Dysmenorrhea
- Dosing: Adult : Contraception: Oral: 1 tablet once daily
- Dose Adjustments
- Renal Impairment: no dosage adjustments
- Hepatic Impairment: contraindicated in patients with hepatic impairment.
- Adverse Drug Interaction:
Headache, migraine, Nausea, vomiting, Nipple pain, depression, emotional lability, and mood changes. - Moreover, hypertension, venous thromboembolism
- Pharmacodynamics/Kinetics:
- Firstly, Half-life elimination: EE: 10-16 hours, NGMN: 18-25 hours, NG: 38-45 hours
- Secondly, Metabolism: Hepatic
- Thirdly, Excretion: Urine and faeces
- Important Notes:
- Firstly, cigarette smoking increases the risk of serious cardiovascular events from combination oral contraceptive (COC) use.
- In addition, this risk increases with age, particularly in women older than 35, and with the number of cigarettes smoked.
- For this reason, COCs are contraindicated in women who are older than 35 years and smoke.
- Pregnancy & Lactation: Use is contraindicated in pregnant women/ may be present in breast milk
- Medication Safety issue:
- Firstly, Ortho-Cyclen may be confused with Ortho-Cept
- Secondly, Ortho Tri-Cyclen may be confused with Ortho Tri-Cyclen Lo

Read More:
Copyright ©: All content on FADIC Website, including medical opinion and any other health-related information, and drug Informtation is for informational purposes only

 Log in
Log in Sign up
Sign up